
Image credit: Eva-Mary Crone, via Pinterest
[English]
[Korean]
Generate by NotebookLM
This week, many people talked about NewLimit after it raised $130 million. The startup, co-founded by Coinbase CEO Brian Armstrong, works on epigenetic reprogramming.

If you’re reading this newsletter, the headline may have felt familiar.
This company focused on something we’ve been exploring together for a while now: epigenetics.
This episode, we’ll take a closer look at the science behind epigenetic reprogramming in aging—and how a new wave of biotech startups is working to turn that science into real-world therapies.
A Turning Point in Epigenetic Science
The Birth of iPSCs (2006)

The Nobel Prize
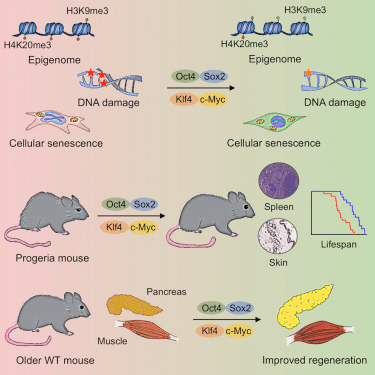
In 2006, Dr. Shinya Yamanaka stunned the scientific world by showing that just four transcription factors – Oct4, Sox2, Klf4, and c-Myc – could revert adult cells into a stem cell-like state.
This was the birth of induced pluripotent stem cells (iPSCs), and with it, the powerful idea that cellular identity could be reprogrammed by targeting epigenetic marks.
It demonstrated that by reversing a cell’s epigenetic state–namely, DNA methylation and histone modifications–cells could be reprogrammed.
This offered an ethical alternative to embryonic stem cells (ESCs) and proved that a ‘biological reset button’ for cell fate was possible. This breakthrough earned Yamanaka the Nobel Prize in Physiology or Medicine in 2012.
If the term sounds familiar, we explored this connection between iPSC technology, regenerative potential, and the future of biotech in a previous episode 📑 : Sci-Fi Is Becoming Real: Are You Mind and Body Ready?
The Rise of Partial Reprogramming (2010s)
In Vivo Amelioration of Age-Associated Hallmarks
by Partial Reprogramming, 2016, Cell
It didn’t take long before researchers began asking: if we can erase cellular identity, can we also reverse cellular age without losing function?
This sparked a decade of exploration into partial reprogramming– using OSK factors to roll back biological age markers while preserving cellular function and without triggering tumors.
Early studies by Juan Carlos Izpisua-Belmonte showed that intermittent OSK expression in mice could enhance regeneration and extend lifespan, all without triggering tumors.
In vivo restoration of function (2020s)
Loss of epigenetic information as a cause of mammalian aging, 2023, Cell
Then in 2020, Dr. David Sinclair’s lab at Harvard demonstrated that partial reprogramming could restore vision in aged mice.
In follow-up work, the team also applied this strategy to the entire body of aged mice, showing improved function and resilience across multiple tissues, including muscle and brain – further supporting the hypothesis that aging is driven by information loss in the epigenome.(Cell, 2023)
This work offer that that aging in mammals could be functionally reversed using epigenetic tools.
The Biotech Acceleration (2020s–)
Since the breakthrough in epigenetic reprogramming, interest in the field has rapidly accelerated. Research groups are racing to refine OSK-based protocols, while biotech startups are leading the charge to translate these discoveries into safe and effective therapies.
Notable players include Altos Labs (backed by Jeff Bezos with $3 billion in funding), Retro Biosciences (which raised $180 million from OpenAI CEO Sam Altman in 2023 and is reportedly pursuing a $1 billion Series A), and NewLimit, all aiming to bring age-reversing therapies to the clinic.
Together, these ventures reflect a pivotal shift: the future of aging science may be shaped not only by academia but by ambitious biotech startups determined to rewrite the rules of biology
What Still Needs Solving – And Who’s Working on It
Despite remarkable progress, epigenetic reprogramming still faces major scientific and practical hurdles:
• Safety: Uncontrolled reprogramming can lead to tumor formation. Companies like Altos Labs and Rejuvenate Bio are exploring refined protocols – Partial Reprogramming – remove oncogenic factors (like c-Myc) or control OSK expression timing.
• Tissue specificity: Ideally, reprogramming should only occur in targeted organs or cell types. NewLimit is developing epigenetic assays and delivery systems to better guide where interventions happen.
• Delivery: Safely and efficiently delivering reprogramming factors (e.g., OSK) remains a challenge. Some startups are testing viral vectors, while others explore non-viral options like mRNA or protein delivery.
• Functional validation: Many interventions show epigenetic changes – but whether those translate to restored function is still being tested. Labs like Sinclair’s are working to link methylation reversal with actual tissue rejuvenation.
Progress is real, but so are the open questions.
That’s why this field remains one of the most exciting and closely watched frontiers in aging science.
Beyond Reprogramming: Slowing the Clock While Science Advances
While epigenetic reprogramming is one of the most promising frontiers in aging science, it’s also still maturing.
But here’s the good news:
As our understanding of aging deepens, we’re also learning how to intervene earlier and more gradually – not just in theory, but in practice.
Even without resetting cells entirely, we now know that it’s possible to slow the pace of aging by targeting the mechanisms that drive epigenetic drift.
And it’s not just scientists leading the charge.
An increasing number of people – from researchers to early adopters – are integrating these interventions into real life.
This growing clarity is shaping a new phase of longevity:
The science and self-experimentation are evolving together.

Leave a comment