
Last week, we explored epigenetic reprogramming. While it’s one of the most promising frontiers in aging science, it’s still a developing field.
But here’s the good news:
As our understanding of aging deepens, we’re also learning how to intervene earlier and more gradually – not just in theory, but in practice.
[English]
[Korean]
Generated by NotebookLM
Even without resetting cells entirely, we now know that it’s possible to slow the pace of aging by targeting the mechanisms that drive epigenetic drift.
And it’s not just scientists leading the charge. An increasing number of people – from researchers to early adopters – are integrating these interventions into real life.
This growing clarity is shaping a new phase of longevity: one where the science and the self-experimentation are evolving together.
Before diving into the real-world strategies, it’s helpful to map where the biological breakdowns are happening – and which interventions are being developed to address them.
The table below summarizes six key epigenetic failure points and highlights the current approaches being explored to target each one.
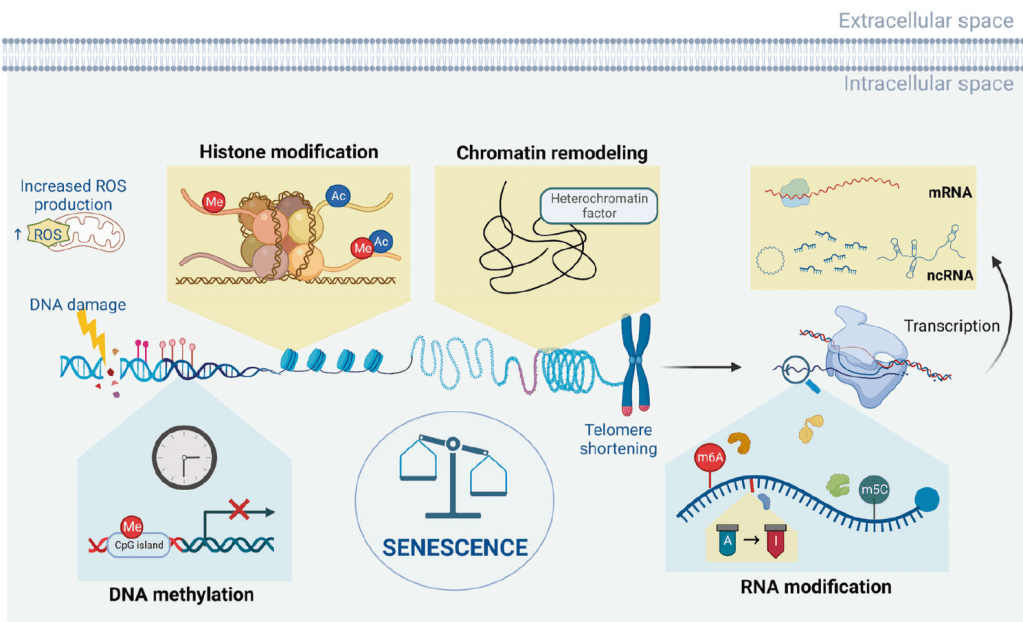
An overview of the aging epigenome. During aging and the emergence of cellular senescence, a series of epigenetic changes occur in cells, including alterations in DNA methylation, chromatin remodeling, histone modification, RNA modification, and ncRNA regulation

Although we’ve laid out the studies in a table to make things easier to follow, each intervention actually affects multiple stages of the epigenetic process in intertwined ways.
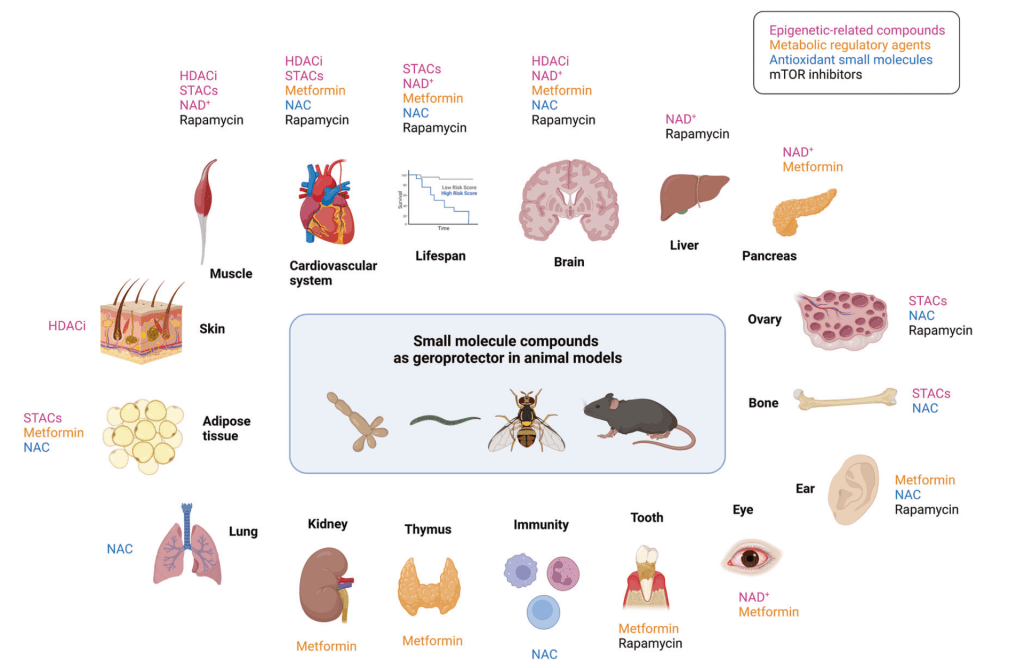
Small molecule compounds as geroprotectors in diverse animal models
From Targets to Tactics
If the table above showed where aging breaks down, the sections below explore how different intervention types are being applied.
We can group these real-world approaches into four categories: drug repurposing, bioactive supplementation, early-stage experimental therapies and lifestyle modification.
1. Drug Repurposing

Originally developed for heart disease,
Viagra found new life
Using FDA-approved drugs for new, aging-related purposes.
This approach is especially attractive in the aging field because it builds on drugs with decades of safety data and large-scale real-world outcomes. Given the slow, cumulative nature of aging, these well-characterized compounds offer a rare opportunity: potential disease prevention or delay at relatively low risk.
• Metformin: With over 60 years of clinical use for type 2 diabetes, metformin reduces glucose production and improves insulin sensitivity, both tied to age-related metabolic decline. It also activates AMPK and reduces mTOR signaling – both associated with longevity. It is one of the most widely self-experimented longevity drugs in the world, used regularly by scientists like David Sinclair and Nir Barzilai, as well as a growing number of physicians and biohackers in low doses.
The TAME trial (Targeting Aging with Metformin), led by Dr. Nir Barzilai at the Albert Einstein College of Medicine, will enroll 3,000 adults to test metformin’s impact on age-related disease onset across multiple domains. It plans to track rates of heart disease, dementia, cancer, and death in people taking metformin versus those who aren’t. But due to funding delays, the study hasn’t fully launched, and early results are still years away.

May 1, 2025 @ NYT
• Rapamycin: Originally approved for organ transplant recipients, rapamycin is a potent mTOR inhibitor – a pathway long linked to lifespan in animal models. It improves immune function and extends lifespan in mice. A 2024 real-world longevity study led by AgelessRx is now testing its bioavailability, safety, and metabolic impact in healthy adults (AgelessRx study). Used by: Matt Kaeberlein and Peter Attia. Bryan Johnson recently stopped after five years of experimentation.

Sept. 24, 2024 @ NYT
• GLP-1 receptor agonists: Initially developed for diabetes and obesity, GLP-1 drugs like semaglutide are now under investigation for their ability to reduce systemic inflammation, improve vascular health, and enhance cognitive resilience. A widely cited systematic review and meta-analysis linked GLP-1 use to reduced all-cause mortality and cardiovascular events. Off-label use in low doses is spreading across longevity clinics.
2. Supplementation
Bioactive compounds people are using to support epigenetic health and cellular resilience.
• HDAC inhibitors: Histone deacetylases (HDACs) regulate chromatin accessibility and gene expression. Natural HDAC inhibitors include butyrate (a short-chain fatty acid produced by gut microbiota), sulforaphane (found in cruciferous vegetables), and curcumin. Pharmaceutical HDAC inhibitors are also being studied for cognitive aging and cancer-related epigenetic reprogramming.
• N-acetyl-L-cysteine (NAC): A precursor to glutathione, one of the body’s most important antioxidants. NAC helps mitigate oxidative stress, supports mitochondrial health, and may help slow cellular aging by reducing ROS-induced DNA damage. It’s also being studied for its role in cognitive support, inflammation reduction, and epigenetic stability in neurodegenerative and age-related conditions.
• Sirtuin-activating compounds (STACs): Sirtuins are a family of NAD+-dependent enzymes involved in regulating metabolism, DNA repair, inflammation, and longevity. Resveratrol, a polyphenol found in red wine, is one of the best-known STACs, shown to activate SIRT1 in preclinical studies. Though human data remains limited, it remains popular for its synergy with NAD+ boosters.
• NMN / NR: NAD+ precursors that enhance energy metabolism, sirtuin activity, and DNA repair. A randomized, placebo-controlled trial led by Dr. Shalender Bhasin at Brigham and Women’s Hospital is evaluating NMN’s impact on exercise recovery, cognitive performance, and physical stress resilience using a formulation from MetroBiotech.
• Fisetin / Quercetin: Plant-derived senolytics capable of selectively clearing senescent cells in animal studies. Fisetin is currently being tested in the AFFIRM trial (NCT04771611) for its effects on inflammation, mobility, and cognition in older adults. Researchers have also explored a synthetic combination called D+Q (dasatinib + quercetin), which has shown synergistic senolytic effects in mice and early human studies targeting idiopathic pulmonary fibrosis and other aging-related conditions.
• TA-65: Derived from astragalus root, TA-65 is thought to activate telomerase, potentially slowing or reversing telomere attrition. A small human study (Harley et al., 2011) reported improved immune profiles and modest telomere lengthening. While evidence remains early, it is a mainstay in longevity clinics.
3. Other Experimental Therapies
Still early-stage, but gaining traction.
• Plasma exchange / dilution: Inspired by parabiosis studies in mice, this approach replaces or dilutes aging plasma components. Bryan Johnson briefly tested plasma exchange from his son, later discontinuing it. Some longevity enthusiasts instead opt for routine plasma donation as a lower-risk way to reduce pro-inflammatory plasma components.
• Gene therapies: Follistatin gene therapy for muscle growth is being trialed in small-scale human and preclinical animal studies. Bryan Johnson received an experimental follistatin gene-therapy injection.
• In vivo partial reprogramming: Companies like Altos Labs and Retro Biosciences are testing safe delivery of reprogramming factors (OSK) to roll back cellular age without tumorigenesis – a topic we explored in more depth in last week’s episode.
These approaches remain speculative but are pushing the boundary of what’s possible — from transient epigenetic resets to systemic rejuvenation.
4. Lifestyle Intervention
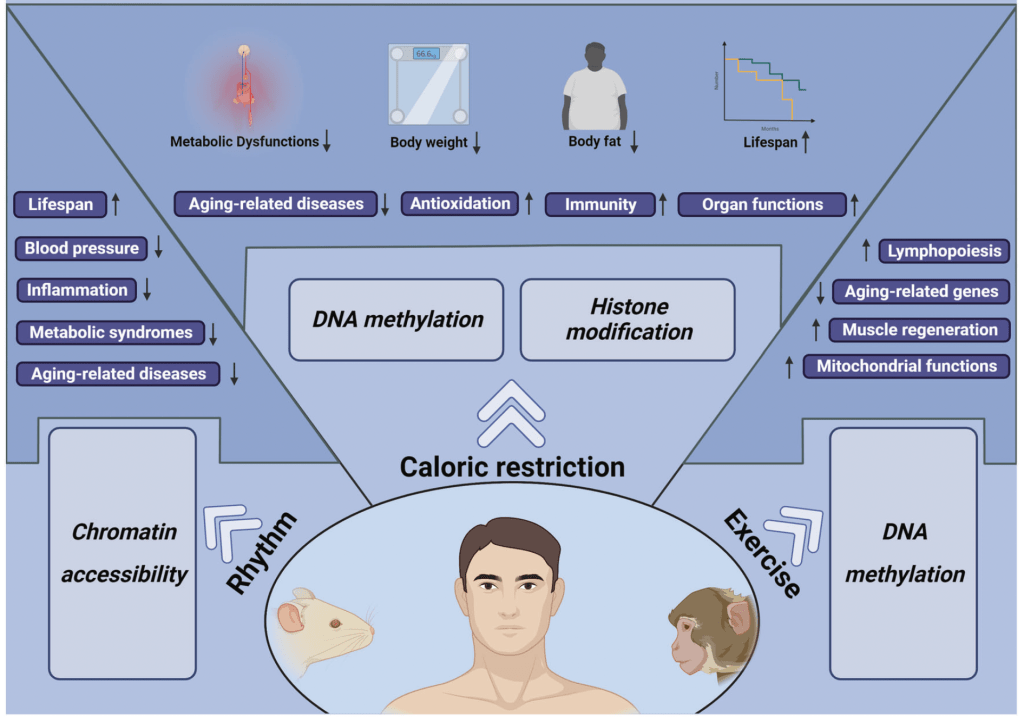
A healthy lifestyle to postpone aging.
The tactics above are still being explored.
Many haven’t been fully proven safe in humans, and potential side effects are still being studied. We also need more research on their long-term use and preventive benefits in healthy individuals.
If you’re wondering where to start, begin here.
Among all interventions, lifestyle change offers the strongest safety profile, the lowest barrier to entry, and the clearest link to slowed biological aging.
Emerging epigenetic science continues to affirm that your daily routines – what you eat, how you move, when you sleep – directly shape the molecular instructions your cells follow.
Lifestyle interventions are not only the most accessible, but also some of the most powerful tools for modulating gene expression, maintaining chromatin structure, and slowing methylation drift over time.
• Time-restricted eating: Aligning food intake with circadian rhythms has been shown to improve metabolic health and reduce epigenetic noise. Animal and human studies suggest benefits for inflammation, insulin sensitivity, and even DNA methylation markers.
• Exercise: Regular physical activity – especially aerobic and resistance training – promotes favorable shifts in histone acetylation, supports chromatin remodeling, and reduces systemic inflammation. One study found that exercise can reshape DNA methylation patterns in muscle tissue, enhancing longevity-related pathways.
• Anti-inflammatory diet: Diets rich in polyphenols, fiber, and healthy fats, while low in processed sugars and refined carbs, correlate with lower biological age scores on DNA methylation clocks. These diets reduce oxidative stress and help maintain histone balance and chromatin structure.
• Sleep & circadian alignment: Sleep is the window when epigenetic maintenance ramps up – including DNA repair, sirtuin activation, and RNA processing. Disruption of circadian rhythms has been shown to accelerate epigenetic aging in both animal models and human shift workers.
What’s Next – and Why You Shouldn’t Wait
Are you waiting for the day a single pill will extend your healthspan?
As we’ve explored throughout this newsletter, the pace of scientific progress in aging is accelerating. But we’re still far from widespread, validated applications.
Even if we do uncover breakthrough mechanisms sooner than expected, deploying them safely and equitably across the general population will be constrained by systemic realities. (You’ve Outlived the System — But It’s Still Running Your Health)
Big pharma must prioritize long-term revenue from existing conditions.
Policymakers must design for population-wide averages.
Physicians must wait for large-scale validation before offering new treatments.
These constraints mean that only those with access to cutting-edge science, a deep understanding of their individual biology, and a willingness to take calculated risks will be the first to benefit.
Boutique longevity clinics already exist and are growing around the world. But for most people, those tools won’t be available for another 10 to 20 years.
So the real question is:
will you wait for the system to catch up?
With the rise of AI-powered research, lifestyle insights are becoming more refined, and access to longevity science is rapidly democratizing.
As a result, self-experimentation – once reserved for elite scientists and fringe biohackers – is now spreading among everyday people.
Or will you begin now – starting with the safest and most science-backed levers – and take agency over your biological trajectory?
We’re building the tools and platform to empower you to do just that.

Leave a comment